Quest Diagnostics Launches Groundbreaking D2C Blood Test for Early Alzheimer’s Detection
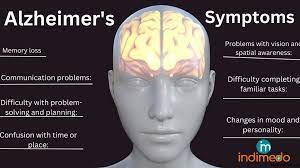
Quest Diagnostics, a renowned name in the medical diagnostics industry, has taken a significant leap forward in Alzheimer’s detection with the introduction of AD-Detect, the first-ever direct-to-consumer blood test to identify abnormal levels of beta amyloid. This crucial Alzheimer’s protein can appear years before dementia symptoms manifest, providing individuals with invaluable early insight into their risk for the disease. The test, available for $399, utilizes the same advanced technology as the blood test previously launched for use by doctors. This groundbreaking development comes in the wake of Leqembi, a drug from Eisai and Biogen, receiving full U.S. regulatory approval earlier this month for its efficacy in removing amyloid from the brain and slowing the progression of Alzheimer’s in early-stage patients.
Empowering Individuals with Early Alzheimer’s Risk Awareness
Dr. Michael Racke, Quest’s medical director of neurology, highlights one of the primary advantages of the amyloid test – it enables individuals to identify their Alzheimer’s risk years before any symptoms appear. This early risk awareness is crucial in planning appropriate preventive measures and seeking timely medical intervention, potentially altering the trajectory of the disease.
Revolutionizing Alzheimer’s Treatment Approaches
Previous Alzheimer’s treatments have primarily focused on managing symptoms, failing to address the underlying disease pathology. However, with Leqembi’s regulatory approval, the landscape is changing. Leqembi has demonstrated its efficacy in removing amyloid from the brain, paving the way for a novel approach to tackling Alzheimer’s disease. Additionally, Eli Lilly’s donanemab is currently under review by the U.S. Food and Drug Administration, promising further hope for patients and their families.
Understanding AD-Detect: A Game-Changer in Alzheimer’s Detection
Quest’s AD-Detect test targets adults aged 18 and older who may have mild memory loss or a family history of Alzheimer’s, offering them the opportunity to comprehend their individual risk for the disease. The test process begins with users paying for the test on Quest’s website. Subsequently, Quest coordinates an appointment with a telemedicine doctor who places the order on the user’s behalf. In case the test yields a positive result, individuals are contacted by a doctor from an independent physician network. The doctor discusses the next steps with the individual and may facilitate sharing the results with other healthcare providers if necessary.
FDA Review and the Future of Direct-to-Consumer Testing
It is worth noting that Quest’s lab-developed test, created and performed in a single laboratory, has not undergone FDA review. However, as long as such tests are prescribed by healthcare providers, the FDA generally refrains from reviewing them. This development opens up possibilities for future direct-to-consumer testing, empowering individuals to take charge of their health and wellness with greater accessibility to advanced medical diagnostics.
Quest Diagnostics’ launch of AD-Detect marks a pivotal moment in the fight against Alzheimer’s disease. This pioneering direct-to-consumer blood test offers a life-changing opportunity for early risk detection, empowering individuals to make informed decisions and seek timely medical intervention. With the success of Leqembi and the potential approval of donanemab, the landscape of Alzheimer’s treatment is witnessing remarkable advancements. While further research and regulatory considerations are essential, direct-to-consumer testing holds promise for democratizing healthcare and fostering a proactive approach to disease prevention and management.